
By the end of 2018 in the United States, there were over 780,000 individuals with kidney failure, also known as end-stage renal disease (ESRD). People with ESRD are unable to filter out waste or manage the levels of fluid and electrolytes in their bodies. Currently, there are only two ways to treat ESRD: receiving a kidney transplant or receiving dialysis. Because there are far more people every year who require a kidney transplant than there are kidney transplant donors, the vast majority of ESRD patients will need dialysis treatment at some point in their care.
Most patients treated with hemodialysis suffer from high blood pressure. Since high blood pressure can lead to poor health outcomes (such as cardiovascular-related events and organ damage), regular monitoring and treatment could lead to better health outcomes.
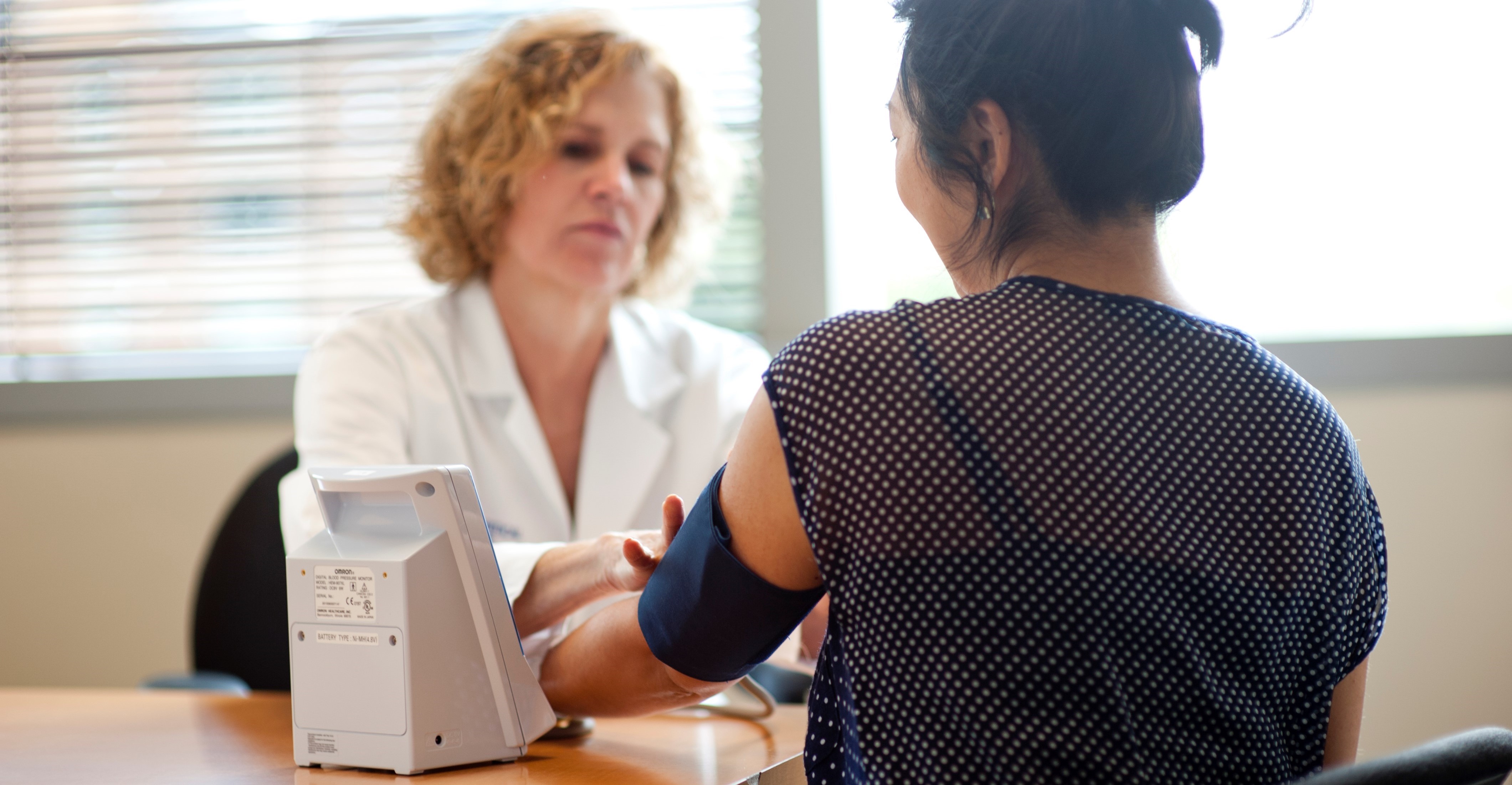
Currently, hemodialysis patients can get their blood pressure taken in different settings, such as the dialysis unit, during clinic visits, or even at home. These different settings may produce different blood pressure readings for the same patient. The current standard of practice is to use blood pressure measurements taken at the start of dialysis when making decisions about treatment. However, recent studies have shown that treating blood pressure based on measurements taken at home may be a better alternative.
The HOME-BP study aims to compare and understand the impact of treating home blood pressure measurements versus blood pressure taken at the dialysis unit. This study will be conducted by teams at both the University of Washington in Seattle and the University of California, San Francisco. By volunteering to participate in this study, you may contribute to improving the standard of care in managing blood pressure among hemodialysis patients.
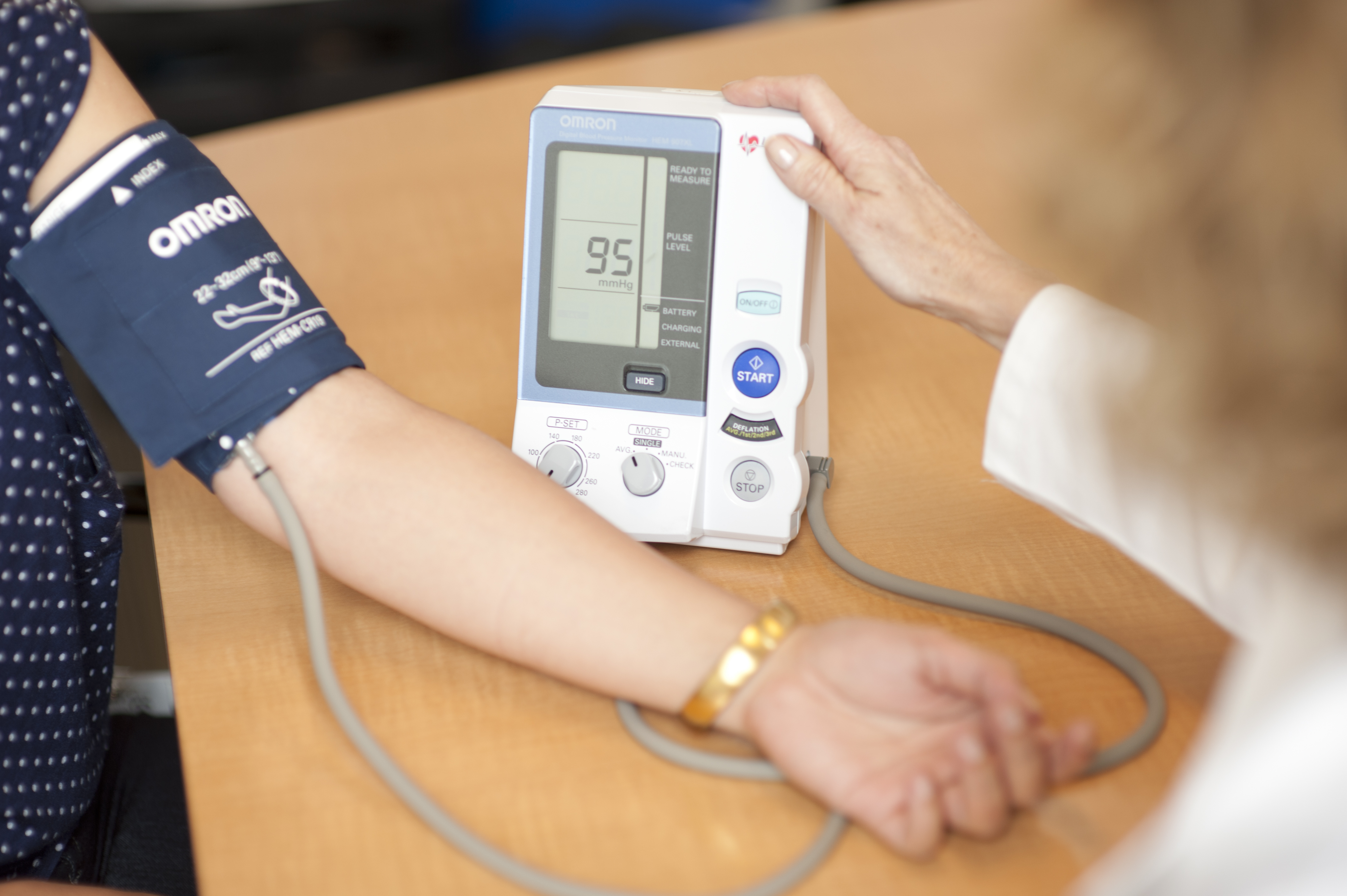
At the beginning of this study, we will give you a blood pressure monitor and training on how to measure, record, and transmit blood pressure readings. You will then take your own blood pressure each week twice on a non-dialysis day in addition to having your usual blood pressure measurements taken at your dialysis sessions. This will continue for the entire 10-month study.
You will be randomly assigned to one of two treatment groups at the beginning of the study, and will transition to the other group after 5 months. You and your doctor will know which treatment group you are assigned to at each part of the study. Your doctor and the clinical research team will use your treatment group to determine how we adjust your dry weight target and your medications to manage your blood pressure. Switching treatment groups will only impact which reading the clinical team uses to make decisions around your blood pressure treatment.
Every 2 weeks, we will meet with you at your dialysis unit to conduct follow-up visits and ask questions about your health, any changes to your medications or medical history, and your recent experiences with cramping/fatigue. We will inform you if any changes to your dry-weight or blood pressure medications have been made based on your blood pressure readings.
You will receive $150 compensation for your participation throughout the study. Additionally, you will have the option to keep both the iPod Touch and the OMRON blood pressure monitor that you were provided, once the study has finished.

Site-specific contact details
University of Washington
Study Principal Investigator: Nisha Bansal, M.D.
Site coordinator: Kaeleb Laszlo |206-720-8524
klasz@nephrology.washington.edu
University of California, San Francisco
Study Principal Investigator: Chi-yuan Hsu, M.D.
Site coordinator: Rose-Ann Thomas |415-425-5098
roseann.thomas@ucsf.edu